I’m considering taking semaglutide in addition to dapagliflozin 10 mg to decrease further my HOMA-IR (from 1.4 to below 1).
Does this make sense?
I saw that others considered or tried GLP1RA + SGLT2 (poke @shc & @AlexKChen ):
A paper published 2 days ago found that “GLT2is + GLP-1RAs combinations were associated with significantly lower all-cause mortality when compared with individual therapies (odds ratio [95% confidence interval [CI] 0.49 [0.41, 0.60]; p < 0.00001).” (Effectiveness and safety of the combination of sodium–glucose transport protein 2 inhibitors and glucagon-like peptide-1 receptor agonists in patients with type 2 diabetes mellitus: a systematic review and meta-analysis of observational studies 2024)
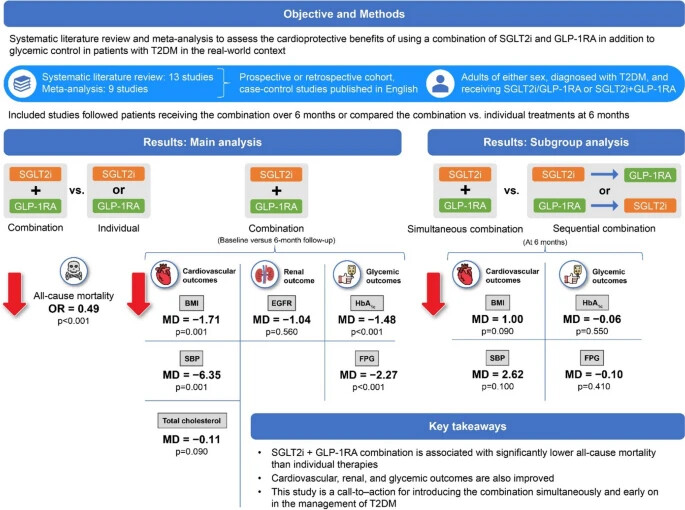
If true, that’s massive. But it’s based on retrospective studies and not RCTs.
Novo Nordisk ran a phase 1 trial of a fixed-dose combination tablet (oral semaglutide + dapagliflozin): A Research Study Investigating How Semaglutide and Dapagliflozin Act in Your Body When Dosed in One Tablet
The trial ended last year. The results haven’t been published. But Novo Nordisk’s Annual Report 2023 says: “Phase 1 trial investigating the effects of the combination of semaglutide and SGLT2i inhibitor dapagliflozin in people with T2D was completed. The project was terminated.” 
Phase 1 is normally for safety and pharmacokinetics. I think that companies are required to report serious and unexpected adverse reactions to the regulatory authorities immediately. So I assume that the project was not terminated due to safety reasons but for commercial reasons? Dapagliflozin was developed by Bristol-Myers Squibb in partnership with AstraZeneca. So maybe Novo Nordisk didn’t want to work with its competitors? 
On the other hand, Novo Nordisk’s FLOW trial of semaglutide in people with T2D and CKD ended early due to efficacy in October 2023, and they’ve just reported “a statistically significant and superior reduction in kidney disease progression as well as cardiovascular and kidney death of 24% for people treated with semaglutide 1.0 mg compared to placebo”. Given that many people with T2D and CKD are already on SGLT2i (and they were not excluded from the trial), I assume that many in this trial were on semaglutide + SGLT2i. Unfortunately, “The detailed results from FLOW will be presented at a scientific conference in 2024.”  As noted by a practician here: “Clinically I cannot wait to see what combination therapy with SGLT2 inhibitors and/or Kerendia will demonstrate.”
As noted by a practician here: “Clinically I cannot wait to see what combination therapy with SGLT2 inhibitors and/or Kerendia will demonstrate.”
There’s also this observational study in a very specific population: GLP-1 receptor agonists-SGLT-2 inhibitors combination therapy and cardiovascular events after acute myocardial infarction: an observational study in patients with type 2 diabetes 2024
The combination of SGLT-2i and GLP-1RA is associated with a reduced incidence of cardiovascular events in patients with T2D and AMI compared with either drug used alone, with a significant effect also on peri-infarcted myocardial rescue in patients without a second event.
The only potential issue (already discussed with @Neo here: New Paper: Summary of the top FDA-approved Drugs that can be Repurposed as Gerotherapeutics / Geroprotectors - #27 by adssx ) is that GLP1-RAs increase insulin secretion, which isn’t great. But do they still increase it significantly in non-diabetics?
(Regarding the cancer risk, this India paper concluded: “Semaglutide use in RCTs and real-world studies was not associated with an increased risk of any types of cancer, and this conclusion is supported by a high grade of evidence.”, Semaglutide and cancer: A systematic review and meta-analysis 2023)
Alternatives don’t seem great:
Are there any other options?