Interesting. I looked at the initial post you had on this and thought I’d missed something as the semaglutide looked best, but then the text said “Thus, it is suggested that GLP-1RAs with greater brain penetrance, such as … may be more likely to modify the clinical course of PD.”
I thought the data above showed the opposite, but figured I’d missed something as I’ve not reviewed the study, but the text seemed to indicate otherwise.
So - if Semaglutide is better on this - I wonder if the trend continues with Tirzepatide or Retatrutide?
Clearly more data needed.
3 Likes
A_User
#232
I’ve heard body weight SNP’s are expressed in the brain, if so, then it’s behavior that leads to lower BMI rather than BMI itself? Those SNP’s might affect neuronal function, so some pleiotropy seem possible here, unlike the SNP’s that just decrease PCSK9 or something.
1 Like
adssx
#233
@A_User: here we have MR + association studies + the experience of practicians pointing to PD patients being quite frail rather than obese.
@DrFraser: that’s the being question. My theory is that because they did not adjust for weight or BMI (it looks like this wasn’t part of their dataset), we cannot conclude, and there might be a confounder due to weight. To add on this: in terms of BBB crossing we know that dulaglutide > exenatide > liraglutide > semaglutide. This paper gives us OR so that the best for PD protection are semaglutide (0.49) > liraglutide (0.70) > exenatide (0.75) > dulaglutide (0.88). So, the exact opposite. And for weight loss, we have semaglutide > liraglutide > dulaglutide > exenatide.
However, we also know that the exenatide phase 2 trial showed improvements in motor and non-motor symptoms. whereas the liraglutide trial only improved non-motor symptoms. That’s why they moved exenatide, but not liraglutide, to a phase 3 trial (results expected in 6 months). There’s an ongoing semaglutide PD trial in Japan. I don’t think anyone is testing dulaglutide, though. So if the phase 2 trials hold, for PD, it seems that exenatide is superior to liraglutide. Which is the opposite of what was found in the above paper.
It also entirely possible that the CI are wide and actually we cannot conclude anything and with more data over a longer period of time (and adjusted for weight) the conclusion will be entirely different.
We can at least say that GLP-1RAs seem neuroprotective, even without weight loss.
A_User
#234
The direction could be like this I would speculate, if not controlled for weight:
Fewer PD genes → More dopamine → Wanting more food → Obesity → Ozempic → (Ozempic decrease PD the most!)
Do you have full access to the article on the GLPs vs. DPP4’s? I need to take a careful read of it.
adssx
#236
I only paid for limited access that prevents me from downloading so the best I can are these screenshots:
4 Likes
Very useful - thank you so much.
adssx
#238
A Mendelian randomization study investigating the causal role of inflammation on Parkinson’s disease 2022
In all the three data sources selected for interleukin-6, we found statistical evidence for an earlier age at onset of Parkinson’s disease associated with increased interleukin-6 concentration [years difference per 1 log-unit increase = −2.364, 95% confidence interval (CI) = −4.789–0.060; years difference per 1 log-unit increase = −2.011, 95% CI = −3.706 to −0.317; years difference per 1 log-unit increase = −1.569, 95% CI = −2.891 to −0.247]. We did not observe any statistical evidence for causal effects of C-reactive protein, interleukin 1 receptor antagonist and tumour necrosis factor α on both Parkinson’s disease and its age at onset.
How do you lower brain IL-6? 
Tocilizumab does not cross the BBB:
IL-6 is part of SASP. Maybe the IL-6 is symptomatic of more cells issuing SASP rather than being causal.
adssx
#240
This MR seems to show that IL-6 is causal, no?
I am not sure how it can.
adssx
#242
I understand that it’s looking at genes associated with high IL-6 only. Not necessarily with high SASP. And high IL-6 only (independently from everything else) is causally associated with a higher risk of PD. Did I misunderstand something?
1 Like
How is high IL-6 causally associated with a higher risk of PD?
A_User
#244
It’s quoted:
In all the three data sources selected for interleukin-6, we found statistical evidence for an earlier age at onset of Parkinson’s disease associated with increased interleukin-6 concentration [years difference per 1 log-unit increase = −2.364, 95% confidence interval (CI) = −4.789–0.060; years difference per 1 log-unit increase = −2.011, 95% CI = −3.706 to −0.317; years difference per 1 log-unit increase = −1.569, 95% CI = −2.891 to −0.247].
Have you missed mendelian randomization studies (and part of the evidence why LDL is causal)?
I have not seen any evidence why LDL is causal of parkinsons.
A_User
#246
I meant the mendelian randomization studies as a method have shown partly why LDL is causal in ASCAD. The same reason why IL-6 is for earlier onset for PD, the method can discover causality.
Why is that relevant in a topic relating to Parkinsons.
A_User
#248
It was pertaining to your question how it is causal with early onset PD, because the MR data shows it.
1 Like
adssx
#249
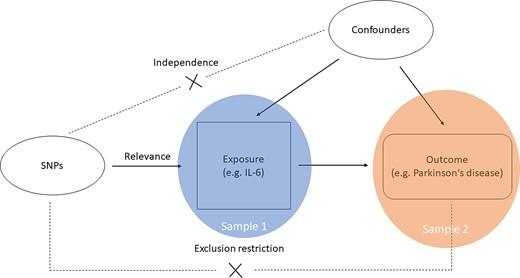
The paper concludes:
We found evidence for a causal association between the onset of Parkinson’s disease and interleukin-6.
In the present study, we explored a possible causal relationship between inflammatory processes and Parkinson’s disease using an MR design, which is a statistical framework that provides valid causal inference on the effect of exposures on outcomes by leveraging genetic information. Connections between inflammation and Parkinson’s disease through that design have been explored by previous studies. We extended this evaluation by considering multiple well-established markers of inflammation. Moreover, we studied the causal effect of the exposures on datasets from different data sources to assess the consistency of the results. Based on the present findings, we did not observe any strong evidence for causality between inflammatory biomarkers and Parkinson’s disease except for IL-6 and AAO.
Of course, there are still many potential limitations.
1 Like
adssx
#250
Risk of Parkinson’s disease divided by 7 among SGLT2 users vs metformin users: Anti-Diabetic Drug Use and Reduced Risk of Parkinson’s Disease: A Community-Based Cohort Study 2024
The study population comprised 86,229 T2DM patients, with 53.9% males. The mean age at the first anti-diabetic drug purchase was 59.0±11.0 and 62.0±11.0 years for men and women respectively. Compared to metformin, several drug types were associated with a significantly lower PD risk: thiazolidinediones (HR=0.91, 95% CI:0.074-1.14); DPP4 inhibitors (HR=0.60, 95% CI:0.53-0.67); meglitinides (HR=0.63, 95% CI:0.53-0.74); GLP-1 agonists (HR=0.54, 95% CI:0.39-0.73); and SGLT2 inhibitors (HR=0.15, 95% CI:0.10-0.21).
Particularly significant in our study is the finding that SGLT2 inhibitors are associated with the most substantial and statistically significant reduction in PD risk.
(I don’t understand why it shows 20 years even though SGLT2i and GLP-1RAs arrive on the market later…)
This confirms that metformin isn’t great for PD (at least at high doses?) and that SGLT2i might be even more neuroprotective than GLP1-RA.
Of note: it seems that only dapagliflozin and empagliflozin are used in Israel as I cannot find canagliflozin here: https://israeldrugs.health.gov.il/#!/byDrug. This paper also says: “any of the available SGLT2is in Israel (empagliflozin or dapagliflozin)”.
Can we find which one is most prescribed in Israel?
5 Likes