adssx
#122
More good news for riboflavin:
-
Association between riboflavin intake and the risk of all-cause mortality of patients with chronic kidney disease: A retrospective cohort study 2024: “After excluding potential confounders, lower intake of riboflavin was associated with the higher risk of all-cause mortality (Q1: HR = 1.33, 95% CI: 1.05–1.69). The similar association was also found in patients at mild/moderate stage (HR = 1.32, 95% CI: 1.05–1.66), in female (HR = 1.35, 95% CI: 1.01–1.81), with hypertension (HR = 1.37, 95% CI: 1.07–1.75), CVD (HR = 1.48, 95% CI: 1.08–2.03), and dyslipidemia (HR = 1.29, 95% CI: 1.01–1.66).”
-
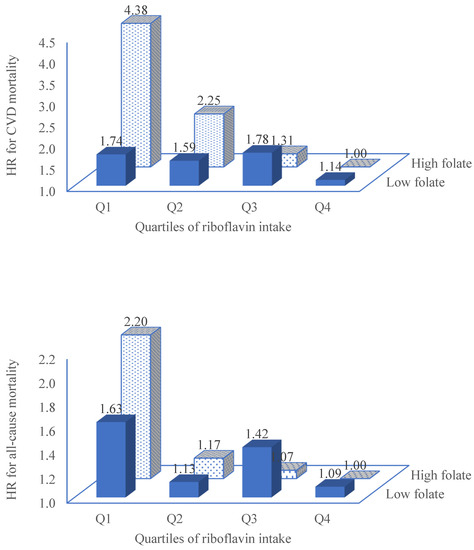
Riboflavin intake associated with decreased risk of all-cause mortality among adults attending NHANES 2005-2016 2023: “High intake of riboflavin was associated with a lower risk of all-cause mortality, and CVD mortality. In multivariable model adjusted for sociodemographic factors, lifestyle factors and chronic conditions, across the quartiles of riboflavin intake, the hazard ratios (HRs) [95% CI] for CVD mortality were: 1.00, 0.92 [0.63, 1.35], 0.79 [0.49, 1.26], 0.52 [0.30, 0.90] (p trend 0.027), respectively. The corresponding figures for all-cause mortality were: 1.00, 0.69 [0.55, 0.87], 0.74 [0.58, 0.94] and 0.62 [0.48, 0.80], respectively. The protective association between riboflavin intake and CVD mortality was further strengthened among those with a high intake of folate (quartiles 3 and 4) with HRs of 1.00, 0.49 [0.21, 1.12], 0.25 [0.10, 0.63] and 0.19 [0.08, 0.47] across quartiles of riboflavin intake (p for interaction 0.039). In conclusion, riboflavin intake was inversely associated with all-cause mortality, particularly CVD mortality. Riboflavin and folate synergistically decreased the risk of CVD mortality.”
-
Riboflavin Intake Inversely Associated with Cardiovascular-Disease Mortality and Interacting with Folate Intake: Findings from the National Health and Nutrition Examination Survey (NHANES) 2005–2016 2022:" Compared to low level (quartile 1, Q1) of riboflavin intake, the hazard ratios (HRs) (95% confidence interval (CI)) for high level (quartile 4, Q4) were 0.53 (0.31–0.90) for CVD mortality and 0.62 (0.48–0.81) for all-cause mortality."
“The mean daily riboflavin intake was 2.1 (SD 1.0) mg ranging from 1.0 (SD 0.2) mg in the lowest quartile (Q1) to 3.4 (SD 1.0) mg in the highest one (Q4).”
As usual, association and not causality, but it’s good that it’s not a U curve.
3 Likes
Good question. I have wondered why homocysteine lowering therapies don’t show clear benefits in clinical trials, especially since I have seen evidence of homocysteine directly harming the vasculature rathern than just being a marker of poor health. The study below indicates that maybe it’s not serum levels that are most important but tissue-bound or intracellular levels of homocysteine. Maybe most homocysteine lowering interventions mainly lower it in the serum but not so much in tissues. I’m not sure, but it’s an interesting theory. Here are a few relevant quotes:
“studies in animals and in cell culture suggest that Hcy has a vast array of toxic effects on the vasculature, with demonstrated roles in endothelial dysfunction, medial remodeling and adventitial inflammation. It is hypothesized that rather than serum Hcy, tissue-bound Hcy and the incorporation of Hcy into proteins could underlie the toxic effects of Hcy on the vasculature.”
" although B-vitamin supplementation successfully decreases serum Hcy concentrations, it is unknown whether it has the ability to decrease tissue-bound or intracellular Hcy. Current evidence suggests that Hcy elicits its negative effects directly in the vessel wall by inducing modifications to vascular cell function, and by irreversibly incorporating into cellular and extracellular matrix proteins (Fig. 2). These pathological changes induced by Hcy would likely be unaffected by serum Hcy-lowering treatments."
Source: Mechanisms of homocysteine-induced damage to the endothelial, medial and adventitial layers of the arterial wall - PubMed
6 Likes
adssx
#124
It seems that LifeExtension was right: Vitamin B12 Levels Association with Functional and Structural Biomarkers of Central Nervous System Injury in Older Adults 2025
Therefore, and since we postulated that people at the lower ranges of B12 may show signs of abnormalities in their mfVEP recordings, we stratified the cohort for B12 levels above and below the mean (geometrical mean = 408 pmol/L). Albeit these B12 levels were considered normal, patients with lower levels of B12 showed a significant inverse association with mfVEP latency (estimate = −0.04; p = 0.023; Fig 2A) in a model correcting for age, sex at birth, CVRF, BMI, APOEε4 allele, HbA1C, and education. Thus, a measured total B12 below the mean, or below 408 pM, significantly associates with a delay in mfVEP latency, representing slower conductivity in the brain.
However, there might be an upper limit (unclear if causal or not or due to a high inactive B12 %):
Surprisingly, in a linear regression model correcting for age, sex, BMI, CVRF, education, APOEε4 status, HbA1C, and creatinine, there was a positive association between Tau concentration (z-score adjusting for the factors stated above) and B12 (β = 0.25, p = 0.005, ΔR2 = 0.052, Fig 4A), but not any of the other biomarkers (Table 3). To further investigate this association between Tau and B12, we investigated the correlation between B12 levels and blood B12 components adjusting for the factors mentioned above. While no association could be found between Holo-TC and any of the included biomarkers (Fig 4B), Tau and UCHL-1 increased with higher Holo-HC (β = 0.22 and 0.28, p = 0.015 and 0.022, respectively, Fig 4C,D). Both associations remained significant after bootstrapping (p = 0.012 and 0.042 for Tau and UCHL-1, respectively).
So until we have more data, the optimal serum B12 range seems to be 400–900 pmol/L? (so 550–1200 pg/mL?)
1 Like
A_User
#125
MMA and HC can diagnose B12 deficiency despite normal B12 levels:
There are two enzymatic reactions that are dependent on vitamin B-12. Vitamin B-12 is required for methylmalonic acid (MMA) to be converted to succinyl-CoA, and in combination with folic acid, for homocysteine (HC) to be converted to methionine [9]. Therefore, MMA is clearly more specific to vitamin B-12 deficiency compared to HC. A deficiency of vitamin B-12 at the tissue level can lead to elevation of both MMA and HC [1] even when serum vitamin B-12 concentrations are within the reference values. Elevated MMA and HC levels together have been found to be 99.8% sensitive for diagnosing functional vitamin B-12 deficiency [10], which is defined as elevated MMA and HC levels despite normal vitamin B-12 levels in asymptomatic individuals [11–13]. Therefore, using serum vitamin B-12 testing alone may under-diagnose the deficiency of this vitamin.
MMA and HC have been identified as early markers of vitamin B-12 deficiency in different subjects such as general population [14], elderly [15–17], Asian Indians [18], infants [19], pregnant women [20], healthy women [21], type 2 diabetes [22], phenylketonuria [13] and renal insufficiency [12]
So I would test MMA + HC + Serum B12.
3 Likes
My levels are 602 pg/mL, I also have not taken B12 very much lately despite being near-vegan
cl-user
#127
That paper is clearly competing with the Chinese ones to reach the lowest quality ever.
we compared a linear with a polynomial regression model. The polynomial model showed a better fit (R2 = 0.023) than the linear model (R 2 = 0.007), suggesting a non-linear association.
I mean seriously? Their models have an R-squared of 0.023 and 0.007, which basically means that there is no meaningful correlation between the model and the data, and they write a paper about it.
Their plots do show that and the funny (or sad) part is that they bravely plot the 95% confidence interval lines while it’s obvious that most of the data points are outside that interval. Look at that one for instance.
4 Likes
adssx
#129
Yes, that’s a problem, but I wouldn’t discard it entirely. UCSF team publishing in the Annals of Neurology, so I give them the benefit of the doubt. I see that paper as a signal (you might say it’s just noise, and you might be right!). Would the trend be more evident with a larger cohort? Only 231 healthy elderly volunteers here, but it’s unlikely we would get all these biomarkers in a large cohort.
1 Like
ok im gonna try activated B6…
Neo
#131
Just take it in reasonable step - unlike some other B vitamins - that you’d to large extent pee out if you have too much:
It’s possible to get too much vitamin B6, primarily from high-dose supplements. Excessive B6 intake, especially over extended periods, can lead to peripheral neuropathy, a condition causing nerve damage and symptoms like numbness, tingling, and burning in the extremities.
2 Likes
FWIW, my homocysteine has gone down from 10 mcmol/L where it’s been steady at that level for many years, to 7 mcmol/L in the year after I started taking TMG 1g/day. I don’t know if it was the TMG specifically, but that’s what I suspect (I also started supplementing with taurine daily at higher doses - for years it was 500mg/day - when my homocysteine was at 10 - to 3-4g/day in the last year or so).
6 Likes
adssx
#133
I think if you take the PSP form of B6 you avoid this risk.
2 Likes
Neo
#134
P5P form? It says activated. I took it just 3x a week and my levels started to shoot up more than I and my doc liked. Is PSP something else?
adssx
#135
Sorry P5P indeed (also called PLP). I think the issue isn’t the blood levels but others forms of B6 self inhibit themselves past a certain oral intake. Otherwise higher is better for B6 serum levels:
Intakes of Folate, Vitamin B6, and Vitamin B12 in Relation to All-Cause and Cause-Specific Mortality: A National Population-Based Cohort 2022
“In men, the multivariable hazard ratios (95% confidence intervals) for the highest versus lowest quintiles of folate and vitamin B6 were 0.77 (0.71–0.85) and 0.79 (0.71–0.86) for all-cause mortality, 0.59 (0.48–0.72) and 0.69 (0.56–0.85) for CVD mortality, and 0.68 (0.56–0.84) and 0.73 (0.60–0.90) for cancer mortality, respectively.”
But the B6 turnover might be more important than the B6 levels themselves and that is something that supplementation could not fix?
with the risks of cardiovascular, cancer, and all-cause mortality in the elderly 2024
“pyridoxal 5’-phosphate (PLP), 4-pyridoxic acid (4-PA), and vitamin B6 turnover rate (4-PA/PLP)”
“Our study found that lower serum PLP levels were associated with increased risks of cardiovascular and all-cause mortality among the elderly population. And higher vitamin B6 turnover rate was associated with increased risks of cardiovascular, cancer, and all-cause mortality.”
I don’t take B6 / P5P but I bought the one you sent to give it a try at some point…
2 Likes
Your levels of what? Hcy?
Neo
#138
Thanks. Don’t know details.
Ok, I don’t know enough, but would make sure to be confident that “ Excessive B6 intake, especially over extended periods, can lead to peripheral neuropathy” is not a risk
—-
Btw, did I recall you saying last year that folate may not be good to get too high? (Even if the all cause mortality above suggest that it might be good to have it relatively high)
adssx
#139
Normally this does not apply to P5P. There is actually ongoing research suggesting that P5P is neuroprotective.
adssx
#140
2 Likes