Bicep
#1212
Acarbose is amazing and I can’t believe you have not tried it yet. Several different and all good modes of action. I always get a gut ache till I start eating the next morning. Go for it.
2 Likes
medaura
#1213
Didn’t you said you’ve tried 500 mg of Metformin? That’s pretty low dose. Why not try to titrate up to 2g a day and see if that makes a dent?
Yes, 500mg/day for one year, zero effects, positive or negative. I have been unwilling to up the dose of metformin, because of the deleterious effect on exercise, and is inferior to exercise in delaying and preventing progression from prediabetes to diabetes. At 66, I am considered an older adult - Metformin inhibits mitochondrial adaptations to aerobic exercise training in older adults:
Quote:
“Metformin and exercise independently improve insulin sensitivity and decrease the risk of diabetes. Metformin was also recently proposed as a potential therapy to slow aging. However, recent evidence indicates that adding metformin to exercise antagonizes the exercise‐induced improvement in insulin sensitivity and cardiorespiratory fitness. The purpose of this study was to test the hypothesis that metformin diminishes the improvement in insulin sensitivity and cardiorespiratory fitness after aerobic exercise training (AET) by inhibiting skeletal muscle mitochondrial respiration and protein synthesis in older adults (62 ± 1 years). In a double‐blinded fashion, participants were randomized to placebo (n = 26) or metformin (n = 27) treatment during 12 weeks of AET. Independent of treatment, AET decreased fat mass, HbA1c, fasting plasma insulin, 24‐hr ambulant mean glucose, and glycemic variability. However, metformin attenuated the increase in whole‐body insulin sensitivity and VO2max after AET. In the metformin group, there was no overall change in whole‐body insulin sensitivity after AET due to positive and negative responders. Metformin also abrogated the exercise‐mediated increase in skeletal muscle mitochondrial respiration. The change in whole‐body insulin sensitivity was correlated to the change in mitochondrial respiration.”
Cardiorespiratory fitness (CRF) is critical for aging people. On balance, when it comes to CRF, fat loss and insulin sensitivity, exercise outperforms metformin, and metformin actually abolishes many exercise benefits. There are many other studies.
Furthermore, exercise as part of a lifestyle intervention outperformed metformin when it comes to delaying or preventing progression from pre-diabetes to diabetes, and this is the whole point of why I’m trying to reverse my prediabetes.
Effects of exercise, metformin, and their combination on glucose metabolism in individuals with impaired glycemic control: A systematic review and network meta-analysis:
1 Like
medaura
#1215
Yes but that’s a generality, you’re an individual. You might need a higher dose to respond. Also you can negate the problems of metformin with muscle response to exercise by pairing it with galantamine. Look up the combo on this forum: it turns metformin’s effects on sarcopenia on its head.
1 Like
Thank you. I will look into it further if my current choices of empagliflozin +/- acarbose are ineffective. At some point everything is on the table, including GLP-1RA agents, though I’m trying to go from less risky to more as I escalate the interventions, hoping to be able to stop before metformin or GLP-1a. It’s a long war, and all is fair in this fight.
2 Likes
medaura
#1217
2 Likes
cl-user
#1218
It’s probably not pre-diabetic if you are insulin sensitive. Just hyper glycemic. You can use a CGM to see how fast your glucose goes down when you eat carbs and verify it’s not pre-diabetes.
For instance here is a small N of 1 experiment I made today:
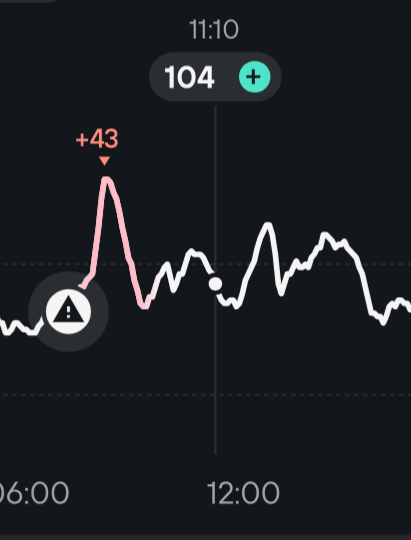
I don’t take anything but black coffee at breakfast but here I also took 2 slices of homemade sourdough French brioche without acarbose and got the +43 mg/dl peak you can see above. It went down very quickly as it should be, so the insulin sensitivity is good. Then around 2:30pm I took the exact same coffee + brioche but with acarbose and got no significative glucose peak.
As I’ve already mentioned I’m in the same category of genetically high blood glucose and tried anything up to and including acarbose and empagliflozin. It makes me send a lot of glucose in my urine but my liver just makes more of it and in the end my A1c stays around 5.9~6.0.
Interestingly it increased my ketones though, which is the mechanism hypothesized to be behind the positive effects of SGLT2i at least for the heart.
Trying Rybelsus right now and the CGM shows the blood glucose is lower. FBG has been lower than 100 for a few days now which is good news new to me.
I think you should consider going straight to a GLP-1a. Next step for me is low dose tirzepatide.
4 Likes
A_User
#1219
Are you measuring your body fat percentage with a DEXA? Your personal fat threshold might be lower than what you have currently making your pancreas unable to work properly leading to prediabetes.
The exercise that will lower glucose after a meal is Zone 2 exercise if I remember correctly, using up glucose. If you have highly dense mitochondria and improved function from doing zone 2 exercise at around 2 mmol/L lactate that might help as well.
If I remember correctly Inigo San Millan have said when his clients start exercising Zone 2 a lot (like 1h 30 min a day) their metabolic parameters improve and they become healthier than they ever been.
1 Like
I do intend to do a dexa reading soon, but I suppose it’s entirely possible I have a low fat treshold, so regardless of the dexa reading I’d like to get my fat % to 10% or so, if it’s higher. Obviously visceral fat I’d like as low as possible.
I would caution you not to jump too fast into GLP-1a, if you read the recent posts in the “experiences with” thread regarding RHR, but my concerns go a lot further than that, especially the more recent ones that also extend into suppressing appetite. Suppressing appetite chemically is very risky business, that has not been adequately - or really much at all - studied. Appetite is stimulated by a variety of hormones and signalling molecules, and is a very strongly evolutionarily preserved function - without hunger, the organism is not motivated to seek energy inputs, and death results. You can bet your bottom dollar that evolution has built up a tremendous physiological infrastructure around hunger and appetite as fundamental to survival. You mess with that at your peril. Already over 20 years ago, I did a deep dive into the physiology of hunger and appetite in the context of CR. I went into more detail in another thread, but hunger signals are tremendously complex, and f.ex. both gherlin and neuropeptide Y are essential for longevity and responsible for triggering most of the CR beneficial effects - interfering with this signalling has been shown in animal models to abolish CR benefits and result in harm. Hunger is an integral part of CR, and exertion is an integral part of exercise. There is a lot more to it, but if I were you, I’d be super cautious about any agents that seek to abort the hunger signal - the consequences could be far reaching. Obviously, do what you feel is right, but research the subject - note how little actual studies there are on the long term effects of suppressing the hunger/appetite signalling cascade compared to the short term effects of weight loss. Losing weight because you eat less and exercise more is absolutely not the same thing physiologically, as eating less because you have no appetite as a result of chemical inhibition and then losing weight.
3 Likes
I think this part needs emphasising more than just a like.
A lot of the appetite suppressants result in relatively easy fat and muscle loss. The fat is easy to replace, but not the muscle.
1 Like
adssx
#1222
Chinese paper (but Peking U), MDPI: Genetic Variation in Targets of Antidiabetic Drugs and Amyotrophic Lateral Sclerosis Risk 2024
Genetic variation in SGLT2 inhibition targets was associated with lower risk of ALS (odds ratio [OR] = 0.32, 95% CI = 0.14–0.74; p = 0.008). We did not find that genetic variation in metformin targets was associated with ALS (OR = 1.61, 95% CI = 0.94–2.73; p = 0.081). Nevertheless, mitochondrial complex I, a target of metformin, was associated with a higher risk of ALS (OR = 1.83, 95% CI = 1.01–3.32; p = 0.047). The analysis showed that genetic variation in sulfonylureas, GLP-1 analogues, thiazolidinediones, insulin or insulin analogues targets was not associated with ALS (all p > 0.05).
mccoy
#1223
The above is interesting because I reached a similar conclusion.
I tend to have a not-low, sometimes approaching 100 mg/dL, FBG. But low homa-IR; in fact, my peaks from fruit or fruit juice on a CGM are just impulsive, very sharp signals, that si, of very low duration.
I wonder, without any scientific evidence, if moderately high FBG may be a normal feature in some individuals. 4 years ago, for example, I was exercising intensely, cardio + resistance and reached my highest bodyweight (muscle+adiposity) and my highest fasting blood glucose (106 mg/dL).
I got worried and turned my diet into a low-carb regime, but the hyperglycemia might have been a normal physiological mechanism to elevate insuline in the body and stimulate IGF-1 for skeletal muscle growth. Unfortunately I went no further with the regime, reduced drastically carbs and lost 10 kg in the process. On hindsight, I wish I kept on, I might have intervened only at higher values, like the diabetic threshold of 125 mg/dL.
A_User
#1224
Worth considering when inhibiting SGLT1 that it seems to make oral rehydration solutions not work in diarrhea (which would lead to dehydration and possibly death):
Oral rehydration solutions (ORSs) is the key treatment of acute diarrhea in children, as it restores the electrolyte balance by stimulating the intestinal sodium/glucose transporter SGLT1 to induce fluid absorption.
https://www.nature.com/articles/s41598-020-64818-3
1 Like
adssx
#1225
Correlation between the utilization of sodium-glucose cotransporter-2 (SGLT2) inhibitors and the risk of dementia: a nationwide population-based study in Taiwan 2024
The impact of sodium-glucose cotransporter 2 inhibitors (SLGT2i) usage on reducing the risk of dementia remains uncertain. Our research seeks to establish the association between dementia risk and SLGT2 inhibitors among individuals with diabetes. This study relied on data from the Taiwan National Health Insurance Database (NHIRD), which was established in 1995 coinciding with the launch of the National Health Insurance (NHI) program by the Taiwanese government. The NHI program was implemented to enhance the healthcare system and public health in Taiwan. Patients with type 2 diabetes mellitus (T2DM) administered SGLT2i between 2016 and 2019 were included in the SGLT2i cohort. The comparison cohort consisted of patients who did not receive SGLT2i, propensity score matching by sex, age (in 5-y intervals), index date year, insurance fee, urbanization, comorbidities, and medications, with a 1:1 ratio of the exposure group. SGLT2i users had a significantly lower risk of dementia than non-SGLT2i users after adjusting for age, sex, insurance fees, urbanization, comorbidities, and medications (adjusted HR = 0.53, 95%CI: 0.50–0.57). The results revealed that patients treated with SGLT2i have a lower risk of dementia in Taiwan.
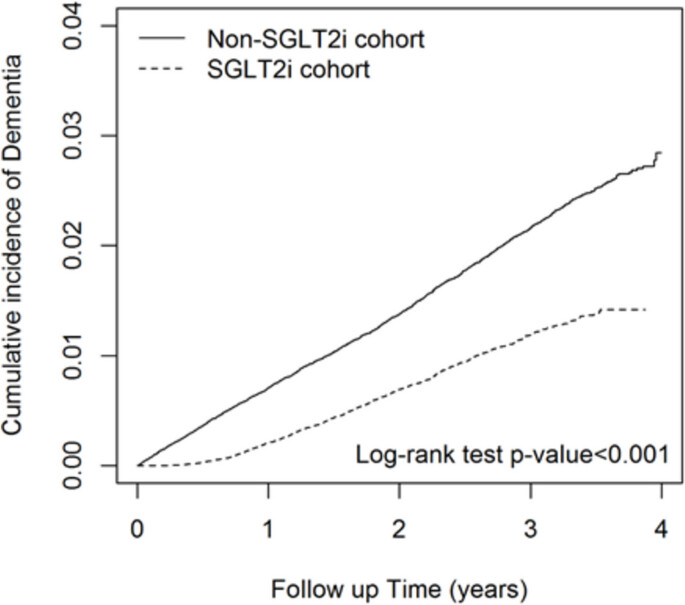
Curves separate too early: might be confounders.
8 Likes
From the study:
“In numerous studies, the dementia prevalence rate in patients with type 2 diabetes was higher than in patients without type 2 diabetes.”
SGLT2i users in their study had lower rates of dementia across age groups. But how much higher are the dementia rates among T2DM patients compared to nondiabetics in Taiwan. The question is whether the SGLT2i users had lower rates of dementia compared to nondiabetics, not just diabetic non users of SGLT2i meds.
1 Like
adssx
#1227
This is not “the” question.
The increased risk of dementia among people with T2D vs non T2D is about 60%. And SGLT2i users with T2D have a 0.6 risk of dementia vs non users. So theoretically we have 1.6x0.6=0.96, so potentially a tiny bit lower than non diabetics not using SGLT2i. “The” question is whether the HR is the same among pre-diabetics and normoglycemic people. I wish they could look at dementia rates among CKD patients as well.
If the rate is significantly lower with SGLT2i despite their minor glucose-lowering effect it points to potential off target effects compared to other diabetes drugs that are more potent.
3 Likes
OK, but they do speculate about the dementia lowering effect of SGLT2i users being down to the SGLT2i not merely as associated with the glucose lowering effect, otherwise any glucose lowering meds should have the same dementia lowering effect. Instead, they speculate that it’s the totality of the SGLT2i actions, including the effect of lowering BP. Also SGLT2i as compared to other T2DM med classes.
The title itself says “correlation”. But assuming a causative effect, is it the same for nondiabtic users of SGLT2i meds vs dementia, because the other potential factors of for example BP are present in both populations, even if at different rates. One would have to also disentangle the effects of BP meds in both.
1 Like
Metformin and exercise don’t mix. What about SGLT2i (dapagliflozin) - not great:
https://academic.oup.com/jcem/article/104/6/1953/5262416
Quote:
"Conclusion
The efficacy of combining two beneficial antidiabetes interventions, regular endurance exercise and SGLT2 inhibition, was not supported. SGLT2 inhibition blunted endurance exercise training–induced improvements in insulin sensitivity, independent of effects on aerobic fitness or body composition."
Note: sedentary, obese subjects.
4 Likes
LukeMV
#1230
Kind of a bummer that there’s evidence that Metformin, Acarbose, and SGLT2’s all have negative effects on exercise
4 Likes
I tried it for months hoping I would adapt to the digestive issues eventually. No luck. Can’t tolerate it.
2 Likes