adssx
#1048
From what I remember the article doesn’t give any quantitative threshold so is fairly useless, no?
1 Like
mccoy
#1049
adssx, I don’t know, I’ve been waiting for such a quantitative model for a while, so I dived down immediately, maybe recklessly, but I’m exploring the article in the other thread, the premises (pathology driven by cumulative exposure) are conceptually compelling.
Edit the above: the article is not on thresholds, rather on a quantitative model of cumulative lifetime exposure to atherosclerosis-producing cholesterol particles.
The article presents a few threshold values, like 1% lifetime excess risk and 10% lifetime excess risk.
There are no guidelines yet nor consolidated procedures on managing this risk, although the conclusion is that an early management (decrease LDL-C as much and as early as possible) ensures a sufficiently low risk later in life.
adssx
#1050
I agree that the concept is compelling but after reading it last time I thought it didn’t move the needle much in the debate and didn’t provide much data, unfortunately.
1 Like
Here’s an interesting paper on arteriosclerosis. They run simulations on arterial plaque buildup. According to their simulations, if your LDL is below 70 and HDL is 60, you will have plaque regression. This sets a target of LDL 70. For me, I’m going to set my target at LDL and ApoB of 50 with an HDL of 50.
Progression model
Regression model
2 Likes
adssx
#1052
It’s a paper from 10 years ago. Surely if LDL < 70 was sufficient to have plaque regression we would have many more recent papers confirming this finding?
5 Likes
adssx
#1053
I found these recent good-quality sources:
-
What Guidelines Say About Risk Reduction: Major Data on the Link Between Lipid Lowering and Outcomes 2024, Canadian Journal of Cardiology: “In meta-analyses of intravascular imaging studies, plaque regression and increases in fibrous cap thickness start to become apparent at an achieved LDL-C level of < 1.8 mmol/L and consistent plaque regression likely requires even lower LDL-C levels (< 1.4 mmol/L). […] Recent evidence shows that achievement of low LDL-C or ApoB (< 1.8 mmol/L or 0.7 g/L, respectively) and very low LDL-C and ApoB (< 1.4 mmol/L or 0.6 g/L, respectively) levels regress and stabilize atherosclerotic lesions on intravascular imaging.”, citing The effects of lipid-lowering therapy on coronary plaque regression: a systematic review and meta-analysis 2021, Scientific Reports: “Our meta-analysis suggests that not only should LDL be reduced to a target level of < 80 mg/dL, but HDL should be increased to a target level of > 45 mg/dL to regress coronary plaques.”
-
Atherosclerotic plaque stabilization and regression: a review of clinical evidence 2024, Nature Reviews Cardiology: “reducing levels of atherogenic lipoproteins, mainly serum LDL-cholesterol (LDL-C), to very low levels can safely reduce overall atherosclerotic plaque burden and favourably modify plaque composition” (which levels though?!)
-
Atherosclerotic coronary plaque regression from lipid-lowering therapies: A meta-analysis and meta-regression 2024, American Journal of Preventive Cardiology: “When comparing PAV reduction by LDL-C levels on follow up (<70 mg/dl vs ≥ 70 mg/dl), both subgroups showed significant PAV reductions, without any significant difference between the groups (difference: 0.57 %; p = 0.37) […] When comparing TAV reduction by LDL-C levels on follow up (<70 mg/dl vs ≥ 70 mg/dl), both subgroups showed significant TAV reduction, but the lower LDL-C (<70 mg/dl) subgroup was observed to have a slightly higher TAV reduction, but this was not statistically significant. (difference: −3.27mm3; p interaction = 0.26) […] This meta-analysis and meta-regression demonstrated the following important findings: 1) use of LLT achieved significant regression of both PAV and TAV; 2) plaque regression was commonly seen in HIS subgroups; 3) both LDL-C level of <70 mg/dl and ≥70 mg/dl at follow-up were associated with a greater reduction in coronary PV; 4) LLT significantly reduced PV and lipid arc, 5) use of LLT significantly increased the FCT. […] In our analysis, both an LDL-C level of <70 mg/dl or ≥70 mg/dl at follow-up wereassociated with a higher reduction in plaque volume. In the previous study by Li et.al where results were stratified by LDL-C level by range, TAV reduction was observed in LDL levels of up to 70–80 mg/dl and PAV reduction was observed even in levels up to 80–90 mg/dl. This suggests that the significant plaque regression in LDL-C levels of ≥70 mg/dl in our study might be due to a slightly higher threshold for plaque regression. […] Moreover, while the addition of ezetimibe seemed to significantly increase the PAV reducing effect of LIS while decreasing the PAV reducing effect of HIS, these inconsistent results were driven by the significant heterogeneity in the LIS plus ezetimibe subgroup (τ2 = 0.88, I2 = 61.8 %), and possibly the limited number of included studies in the HIS plus ezetimibe subgroup. In fact, previous studies demonstrated that the addition of ezetimibe do not contribute to a significant increase in PAV reduction. While the role of statins on plaque regression have been discussed above, we emphasize that HIS was the only LLT subgroup which provided significant reductions in both TAV and PAV. […] While many previous studies have demonstrated that the addition of EPA to statin contributes to increased plaque regression, our pooled analysis showed that the addition of EPA to LIS is superior even when compared to other multiple classes of LLTs, supporting the hypothesis that EPA contributes to plaque regression in mechanisms beyond lipid-lowering, giving emphasis to the role of its anti-inflammatory effects in plaque regression.”
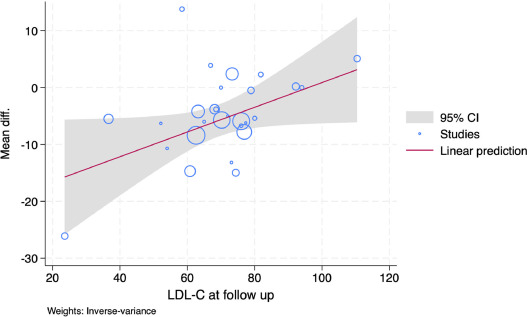
So what matter might be to use statins rather than the LDL level 
5 Likes
It seems that a low intensity stating + ezetemibe+ EPA could also be a winning combination. Heck, throw Bempedoic acid in there as well for good measure.
I did notice that when I added EPA, my triglycerides took a nose dive while my LDL wasn’t much affected.
1 Like
adssx
#1055
For PAV but weirdly not for TAV.
Published today:
Inflammation, Cholesterol, Lipoprotein(a), and 30-Year Cardiovascular Outcomes in Women 2024
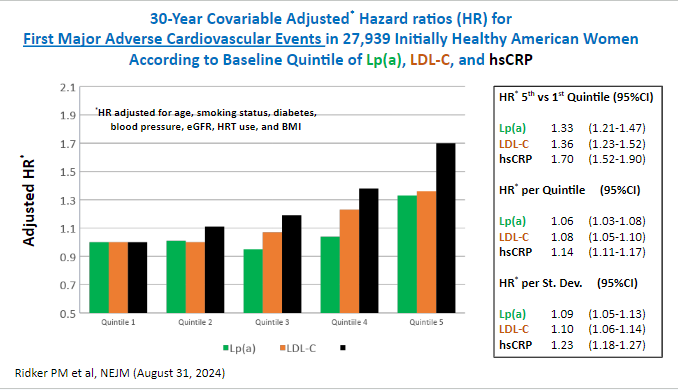
Is hsCRP the most important thing actually?!  If someone can find the values of the quintiles that would be super helpful.
If someone can find the values of the quintiles that would be super helpful.
[EDIT: Actually it’s in women, estrogens might protect against atherosclerosis so LDL might not be as important for them.]
4 Likes
adssx
#1056
Here it is:
Too bad they gave Lp(a) in mg/dL instead of nmol/L: Lp(a): When and how to measure it 2021
Many studies have shown differences between Lp(a) assays. Concerted effort was made by the IFCC to select and characterize a suitable reference material and to develop a multistep standardization protocol to be used by manufacturers and clinical laboratories. However, traceability to the WHO/IFCC SRM-2B improves assay comparability but does not eliminate the isoform sensitivity of the analytical methods.The target value assigned to the reference material is in nanomoles per litre of Lp(a) protein, reflecting a mole for mole interaction of antibody with apo(a). Lp(a) has historically been expressed in mass units (mg/dL) encompassing the mass of the entire particle, including the content of apo(a), apoB-100, cholesterol, cholesteryl ester, phospholipid, triglyceride and carbohydrate. This is metrologically unsound because what is measured by immunoassays is the protein component of Lp(a) and not its lipid and carbohydrate content. Therefore, the most appropriate units of measurement of Lp(a) are nmol/L. Lp(a) concentrations should not be converted from nmol/L to mg/dL, or vice versa, as all conversion factors are inherently isoform dependent.
5 Likes
mccoy
#1057
I’ve expanded the considerations in the specific thread, quantitatively it is possible to estimate one’s own lifetime exposure to ACV events and compare it to the general population and build estimates of the risk development in future given cumulative expected values.
They don’t provide suggested thresholds but we can judge by ourselves, the principle is that the lower the better, possibly below 1%, although that can be impossible to those no more young.
There might be some threshold values in the cited articles, but I’ll have to search it.
1 Like
Are there any issues with HDL being higher than LDL? My father’s latest results are a 34 LDL and 61 HDL. I thought I remembered reading somewhere that one being higher than the other is what is detrimental. Usually HDL is always lower than LDL.
Davin8r
#1060
Is high dose statin therapy also more anti-inflammatory then low dose statins, or is there an anti-inflammatory threshold that is met at lower doses? It’s too bad we don’t have a colchicine group (yet) to throw in there for comparison.
How do you measure “anti-inflammatory” in this context?
adssx
#1062
I don’t think anyone knows yet but the obicetrapib trials will help answer this question. My guess is that it’s okay or even protective.
1 Like
We do know that high HDL can be a negative, but anything in the 60 range is not “high”. Michael Lustgarten has a video somewhere, where he charts at which point HDL becomes a negative, and as I recall, the high end was above the 60’s, although surprisingly not much above 60’s already somewhere in the 70’s.
I am not aware of any data showing HDL higher than LDL being a negative. We can only speculate, and I too would speculate that it would be protective as long as the HDL is not crazy high; at 60-70, that’s not high. But speculation has little value, so ultimately we simply don’t know.
adssx
#1064
I don’t think we know that. There are probably many HDL subtypes with different effects. People who inhibit CETP live long and have low LDL and high HDL: Obicetrapib (CETP inhibitor for dyslipidemia)
2 Likes
That’s why I said “can” be a negative, there are many articles to that effect, and I have definitely heard Thomas Dayspring say so. That doesn’t mean high HDL is always a negative, for some subpopulations it may not… but then again, it’s the same with high LDL - high can be a negative, but not always, because there are documented sky high LDLs in FHC cases who have a normal lifespan with no medication, something in their physiology protects them. Lipids are complicated, full of special cases and exceptions, but general rules exist too. I myself on my last test had an HDL of 92mg/dL, LDL 133, and Lp(a) 78mg/dL (unfortunately, those are the units the lab uses) - and all of this on 10mg atorvastatin. Without the statin, my LDL was in the 180’s and HDLs often over 100. I’ve had very high cholesterol all my life. I’ve only been on a statin for 5 years. And yet, last December, I had a CAC scan, so age 65, and the score was stone cold ZERO. Now, maybe I have a ton of soft plaque, I don’t know, but the cardiologist thinks that’s unlikely and he discouraged an angio, which I wanted. I had a perfectly fine EKG, FWIW. Oh, also, the scan showed:
“The ascending aorta measures 29 mm, which is normal. No aortic valve leaflet calcifications. No atherosclerotic calcifications of the partially visualized thoracic aorta.”
The cardiologist said a 29mm aorta at my age and cholesterol levels is very good.
Point being: with lifelong terrible, unmedicated (until fairly recently) lipids, including high HDL and sky high Lp(a), at age 65, zero evidence of artherosclerosis (at least on a CAC scan). With these terrible numbers I was fully expecting extensive calcifications of my arteries. So: there’s general rules, but always exceptions.
3 Likes
adssx
#1066
“High HDL” is neither negative or positive, as of today, it’s just irrelevant:
And congrats on your 0 CAC score!
4 Likes
adssx
#1068
On plaque regression, does anyone understand his line of reasoning?
And commenting on this paper:
He said:

2 Likes