I’m surprised they didn’t mention 53BP1 as a possible longevity effector of NRF2. It contains multiple antioxidant response elements/AREs in its promoter that mediates its induction by NRF2.
data from Targeting of Nrf2 induces DNA damage signaling and protects colonic epithelial cells fromionizing radiation and also see Oncogenic KRAS drives radioresistance through upregulation of NRF2-53BP1-mediated non-homologous end-joining repair
The longevity implications of 53BP1 revolve around its ability to increase the efficiency and accuracy of DNA double-strand break/DSB repair. Ionizing radiation/IR is a common experimental source of DSBs and FDA-approved Nrf2 activator omaveloxolone promotes IR resistance in hematopoietic system, suggesting that it improves the DSB DNA damage response.
The administration of a brief course of RTA 408 treatment, beginning 24 h after lethal doses of radiation to bone marrow, significantly increased overall survival. Importantly, treatment with RTA 408 led to the full recovery of steady state hematopoiesis with normalization of the frequency of hematopoietic stem and progenitor cells. Moreover, hematopoietic stem cells from RTA 408-mitigated mice showed lineage-balanced, long-term, multilineage potential in serial transplantation assays, indicative of their normal self-renewal activity. [ref]
In further support of this, pretreatment with Nrf2 activator bardoxolone methyl was shown to reduce IR-induced chromosomal aberrations (which represent unrepaired DSBs), irrespective of whether the cell is in G1 or S/G2 phase at the time of IR application. [ref]
53BP1 appears to vastly improve the fidelity of homology-directed repair by promoting homologous recombination (which can perfectly repair DSBs) over single-strand annealing (which can produce massive deletions). See 53BP1 fosters fidelity of homology-directed DNA repair
As homologous recombination is especially important in mitotic cells, this would suggest a potential role for 53BP1 in preserving stemness.
Furthermore, AMPK phosphorylates 53BP1 to promote NHEJ pathway of DSB repair. NHEJ is the primary pathway for repairing DSB in both mitotic and post-mitotic cells, and it’s interesting to consider that some of AMPK’s longevity effects are mediated via this pathway. They only showed AMPK to promote NHEJ efficiency, but I suspect this would also promote fidelity, as it reduces the amount of time that DNA ends are exposed to exonucleases and other enzymes.
One important mediator of NHEJ is the Ku heterodimer and Ku80 protein levels vary dramatically among species and are strongly correlated with longevity, but the bowhead whale and its 200yr+ MLS is probably the best evidence of how important efficient and accurate NHEJ is. Due to high levels of CIRBP and RPA2, bowhead whales have extremely efficient and accurate NHEJ.
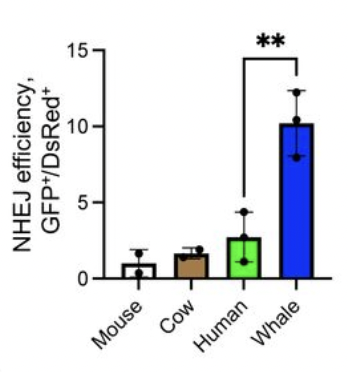
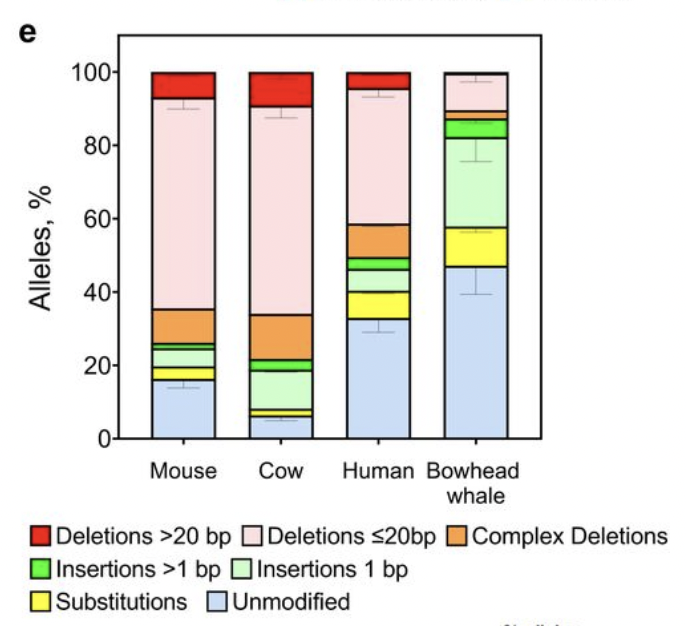
Using CRISPR-induced DSBs you can see that shorter-lived mammals are less likely to repair DSB without modifications, and more likely to introduce large deletions. Whereas bowhead appears to prefer 1bp insertions or unmodified repair. The larger the deletion, the higher probability of cutting into exonic or regulatory sequences. Data from DNA repair and anti-cancer mechanisms in the longest-living mammal: the bowhead whale
Further support for the importance of 53BP1 comes from the paper DNA Damage Detection by 53BP1: Relationship to Species Longevity. They find increased 53BP1 foci formation in response to genotoxic chemicals in longer-lived species, which appears to be due to increased DSB-sensing capacity in longer-lived species.
One last and very intriguing paper (Lamins are rapamycin targets that impact human longevity: a study in centenarians) showed that centenarian fibroblasts have more nuclear prelamin A and that this raises steady-state nuclear 53BP1. Because of this the centenarian fibroblasts can more quickly mobilize DNA repair response in response to oxidative stress-mediated DSBs. And guess what? Rapamycin treatment induced a centenarian-mimetic phenotype, by raising nuclear prelamin A in young fibroblasts which in turn raised nuclear 53BP1. They also showed that a similar effect of lovastatin, which was the first FDA-approved statin and makes up about 2-3% dw of the delicious oyster mushroom.
Maybe I’m just having delusions of grandeur, but I’m imagining that some combination of NRF2 activator (technically most of these are KEAP1 inhibitors), rapamycin, and an AMPK activator might have some special synergy when it comes to maintaining genomic integrity. The NRF2 activator would raise 53BP1 protein levels, the rapamycin would increase its nuclear trafficking, and AMPK phosphorylation would allosterically enhance its activity.