Poor muscle function is increasingly obvious with aging and needs effective and safe medicine for treatment. Trimetazidine (TMZ) has potential benefits for the condition but has not yet been fully recognized. In the randomized-control pilot study part, fifty-three old patients were assigned to the TMZ group or control group. For the TMZ group, a dose of 35 mg of oral TMZ was administered with a meal twice a day for 3 months. Only conventional treatments were administrated in the control group. Muscle strength, gait speed, muscle endurance, and balance maintenance were measured during the visits. In the experiments part, thirty mice were screened and randomly assigned to three groups: model group received a D-gal (500 mg/kg) intraperitoneal injection every two days for six weeks, the control group received saline at the same condition, and the intervention group received 5 mg/kg TMZ solution every two days by gavage for two weeks.
Swimming tests and forelimb grip strength measurements were also performed. Furthermore, significantly clustered profiles from differentially expressed genes were found by RNA-seq and verified by qRT-PCR and WB. Myofiber analyses were done by H&E staining. Here, we found the improvement of skeletal performance in aged individuals and aged mouse. The dominant handgrip strength (HS) was increased by 24.4% and dominant pinch strength (PS) by 12.4% in participants with TMZ modified-release tablets consumption. Exhaustive time was increased by 23.6% and upper limb grip strength by 44.1% in aged mouse with TMZ-treated. Besides, we also identified some newly discovered molecules associated with TMZ on muscle function improvement during aging. To aged C2C12 cells and aged mouse muscle, TMZ-treated was related to a statistically significant decrease in the expressions of NOS3 and MMP-9, but a statistically significant increase in the expressions of OMD and MyoG. In summary, TMZ modified-release tablets can improve the muscle strength of elderly patients. Besides, the improvement of skeletal muscle function affected by TMZ was associated with reducing NOS3 expression in senescent myoblasts.
Bipolar depression is different from major depressive disorder in its ontogeny and clinical characteristics [11, 186]. Their biological differences are highlighted by the fact that antidepressant monotherapy is not recommended to treat bipolar depression [7, 13]. In this review, three major biological processes associated with bipolar depression were highlighted. Overall, evidence for mitochondrial dysfunction, inflammation and oxidative stress in bipolar depression is consistent with many replicated findings across tissue types, molecular assays, and ethnicities [187, 188]. A particularly compelling hypothesis is that bipolar depression is a state of decreased mitochondrial energy generation, which may be overcompensated by increased mitochondrial energy generation in mania [20–25]. Trimetazidine’s main activity in boosting mitochondrial energy generation only when mitochondrial function is reduced, while also targeting inflammation and oxidative stress that occurs in both depression and mania in bipolar disorder makes it a promising novel pharmacotherapy candidate to be tested in clinical trials. Should such trials yield positive outcomes, it can be rapidly translated into clinical care to treat bipolar depression due to its availability, low cost, safety, and tolerability.
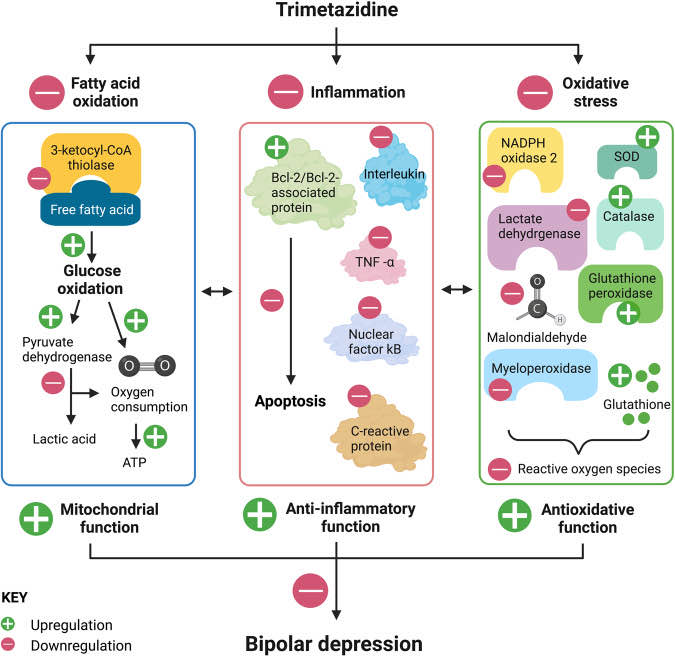
Trimetazidine is a selective inhibitor of 3-ketoacyl-CoA thiolase.
Hence trimetazidine modulates mitochondrial energy production by inhibiting fatty acid oxidation to engage efficient glucose oxidation, which increases adenosine triphosphate (ATP) conversion compared to oxygen consumed. Trimetazidine also increases pyruvate dehydrogenase activity to decrease lactate accumulation. These effects ultimately reduces intracellular calcium ion accumulation and reactive oxygen species (ROS) to reduce apoptosis, inflammation and oxidative stress indicated by reduced level of biomarkers such as tumor necrosis factor alpha (TNF-α) and nicotinamide adenine dinucleotide phosphate (NADPH) oxidase 2. Trimetazidine has also been shown to increase antioxidant activity measured by increased glutathione, glutathione peroxidase, superoxide dismutase (SOD), and catalase. Taken together, accumulating preclinical and clinical evidence of trimetazidine’s regulation of mitochondrial function, anti-inflammatory and antioxidant properties strongly support its potential efficacy to reduce bipolar depression. Figure created with Biorender.com.
It’s available in 
Last, trimetazidine was not associated with any safety issues. There were no statistically significant differences between the treatment groups in the primary safety endpoint. The incidence of adjudicated adverse events was low and well balanced between treatment groups. Notably, a possible association between trimetazidine and Parkinsonism has been previously described. In our study, the occurrence of neurological symptoms such as Parkinson’s disease, atypical Parkinsonism, or drug-induced Parkinsonism, were similar in the placebo and trimetazidine arms.
https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(20)31790-6/abstract