The thing is that it is the reduction in partial pressure that does the job of starting HIF 1 alpha. That can be done at a normobaric pressure, but starting with a higher concentration of Oxygen. I do this using an Oxygen concentrator.
adssx
#373
Hm. This paper still suggests that the oxygen concentration might play a role independent of the pressure:
Impairment of neuronal mitochondrial function by l-DOPA in the absence of oxygen-dependent auto-oxidation and oxidative cell damage 2021
Independent of its function as a precursor for DA synthesis, we demonstrated that l-DOPA can mediate both, a neurotoxic and a neuroprotective activity. Interestingly, the precise kind of action depends directly on the oxygen tension. Under normoxic oxygen levels, auto-oxidation of l-DOPA and linked ROS production prevails, while under hypoxic conditions l-DOPA auto-oxidation is heavily reduced.
So overall our data suggest an oxygen-dependent dual effect of l-DOPA: Under normoxic conditions l-DOPA auto-oxidation produces cytotoxic ROS, whereas under hypoxia and during shorter treatment duration l-DOPA is more stable and the ROS scavenging capacity prevails. Moreover, we observed that neuronal cell lines were much more vulnerable than other human cell types to l-DOPA-mediated ROS.
Oxygen partial pressure will be an issue, but specifcially in getting the HIF 1 alpha nuclear factor initiated it is the change in pressure that makes an issue.
Too much hyperoxia is bad for anyone.
adssx
#375
You wrote “At this point I will assume that switching on HIF from time to time is “a good thing”. Therefore, we need to know how to switch on HIF.” So I’m putting these here without context:
I think HIF 1 alpha is part of the hormesis path.
Apart from exercise I use variations in the partial pressure of Oxygen with a view to systemic stimulation of HIF 1 alpha.
adssx
#377
Some suggest that PGK1 is the rate-limiting step in glycolysis (a process highly impaired in PD): How to accelerate glycolysis and treat Parkinson’s disease 2024
Interestingly, exposing cells to low oxygen conditions can activate hypoxia-inducible factors (HIFs), which can upregulate PGK1 @John_Hemming.
If true, this could explain two PD mysteries:
- Why do symptoms disappear at high altitudes?
- Why do smokers have a lower risk of PD?
Terazosin might do the job: Terazosin / doxazosin / alfuzosin may protect against dementia with Lewy bodies
1 Like
Agetron
#378
More and more we’re finding the importance of the gut biome to the immune system. This latest information on gut biome and Parkinson’s might be of interest to some people.
The fix might be as simple as increasing certain vitamins and supplements like B2 and B7. On a similar vein, for decades ulcers were treated with surgeries that did not prevent the reoccurrence.
We realize that treating with antibiotics can cure certain ulcers. You don’t hear about ulcers the way you use to.
Link: Parkinson's Link to Gut Bacteria Suggests an Unexpected, Simple Treatment : ScienceAlert
3 Likes
adssx
#379
More on vitamin D being probably irrelevant in PD: Associations of milk, dairy products, calcium and vitamin D intake with risk of developing Parkinson´s disease within the EPIC4ND cohort 2024
No relationship was observed between dairy consumption (HR 1.07, 95% CI 0.82–1.39), individual dairy products (milk: HR 0.95, 95% CI 0.73–1.23; yogurt: HR 1.03, 95% CI 0.82–1.29; cheese: HR 1.13, 95% CI 0.85–1.51), or vitamin D (HR 1.08, 95% CI 0.80–1.45) with PD risk. However, we observed a risk-increasing association with higher calcium intakes (HR 1.33, 95% CI 1.00-1.78, p for trend = 0.031), which was more pronounced in men (HR 1.50, 95% CI 1.00-2.25, p for trend = 0.044) and in ever smokers (HR 1.64, 95% CI 1.06–2.53, p for trend = 0.014). No compelling evidence was found for an association between dairy products or vitamin D intake and PD risk indicating a potentially limited relevance of dairy intake in PD risk than previously described. Our observations of a positive association between dietary calcium intake and PD risk in men and in ever smokers require further validation.
This suggests that vitamin D may not be related to the development of PD and that other mechanisms might therefore be responsible for the results in the aforementioned studies on serum vitamin D levels. One Mendelian randomization study explored the association between genetically decreased 25-hydroxyvitamin D concentrations with PD in individuals of European descent from 15 cohorts to investigate a potential causal relationship. The authors concluded that there is a lack of definite evidence supporting a role of vitamin D in PD.
On the other hand, men at risk of PD might prefer to avoid dairy products:
This suggests that vitamin D may not be related to the development of PD and that other mechanisms might therefore be responsible for the results in the aforementioned studies on serum vitamin D levels. One Mendelian randomization study explored the association between genetically decreased 25-hydroxyvitamin D concentrations with PD in individuals of European descent from 15 cohorts to investigate a potential causal relationship. The authors concluded that there is a lack of definite evidence supporting a role of vitamin D in PD.
2 Likes
I think the key role of Vitamin D is to maintain the bodies optional systems that only function when there is more food available. Hence the VDR enables optional genes. These may help with lot of things, but do not prevent diseases of aging.
I think it is clear that PD is a disease of aging as it occurs with age. I start then with the assumption that there is a problem with certain proteins being produced probably as different splices when power levels in the cells go down.
If it is a splicing issue (and there is evidence for this) that would explain a variation between people are different alleles will result in different splices at lower levels.
Hence although I think Vitamin D is someone to keep at a high serum level I don’t think it is an anti-aging vitamin per se.
1 Like
Pender
#381
I know identical twins, age 80. Both have severe arthritis.
One has been on methotrexate and its predecessors for 10-20 years and is mentally sharp. The other was diagnosed with fronto-temporal dementia 5 years ago.
Confounders: twin with dementia was much more sedentary, ate poorly, had untreated hyper-cholesterolemia, had a heart attack in late 40s, had children much later in life, was less social.
2 Likes
adssx
#382
Interesting! A trial of azathioprine (common alternative to methotrexate) has just ended in Parkinson’s disease. Results will be published soon…
3 Likes
adssx
#383
There’s only one old small Brazilian trial of riboflavin in PD: High doses of riboflavin and the elimination of dietary red meat promote the recovery of some motor functions in Parkinson’s disease patients 2003
19 patients, no placebo:
riboflavin orally (30 mg every 8 h) plus their usual symptomatic medications and all red meat was eliminated from their diet
All 19 patients who completed 6 months of treatment showed improved motor capacity during the first three months and most reached a plateau while 5/19 continued to improve in the 3- to 6-month interval. Their average motor capacity increased from 44 to 71% after 6 months, increasing significantly every month compared with their own pretreatment status (P < 0.001, Wilcoxon signed rank test). Discontinuation of riboflavin for several days did not impair motor capacity and yellowish urine was the only side effect observed.
There’s also this ongoing trial: Validation of a Novel Functional Food Designed to Meet the Nutritional Needs of People Living with Parkinson’s Disease (Food4PD). Twice daily: “An enriched functional drink containing chicory inulin (10g), folic acid (100µg), vitamin B12 (5µg), vitamin B6 (5mg), riboflavin (2.5mg) and vitamin D (5µg).”
On the other hand: Dietary intake of folate, vitamin B6, vitamin B12 and riboflavin and risk of Parkinson’s disease: a case–control study in Japan 2010
After adjustment for potential dietary and non-dietary confounding factors, intake of folate, vitamin B12 and riboflavin was not associated with the risk of PD (P for trend = 0·87, 0·70 and 0·11, respectively).
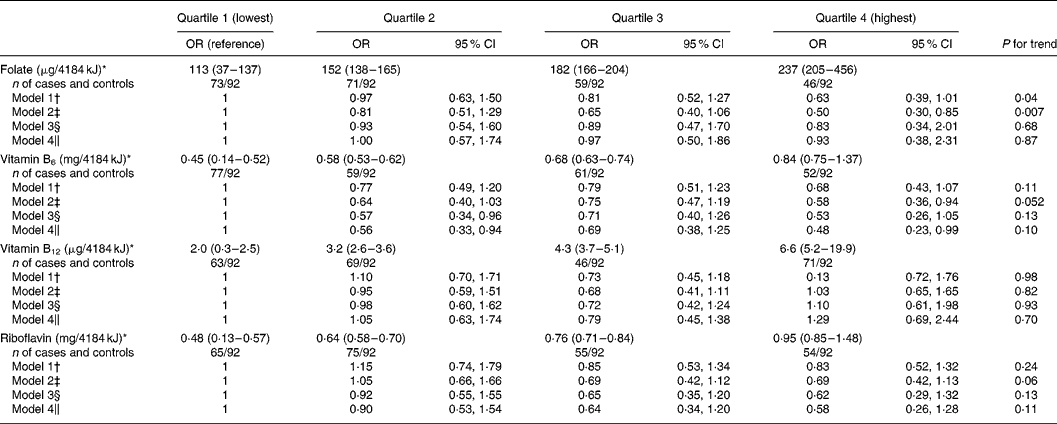
2 Likes
Rapan
#384
At least one epidemiology study suggests that MTX (methotrexate) doesn’t increase risk for Parkinson (for RA patients although RA is correlated with higher risk for Parkinson).
Interestingly, chloroquine and hydroxychloroquine reduced risk by about 25% for Parkinson.
https://www.neurology.org/doi/10.1212/WNL.0000000000013303
2 Likes
Satchel
#385
Perhaps it’s been brought up, but Klotho is mediated through Platelet Factor 4 in the brain and Dana Dubol has shown that PF4 is almost completely protective in PD neurons (not motor neurons). Unlike Klotho, PF4 is readily available for human use for 20+yrs.(used in a surgery setting). Human doses are already published; safety is excellent and I think Dubol was using 1 sq inj every 2-3 wks. I will dig up the paper, but if I had PD I would certainly consider PF4. Lots of different suppliers
3 Likes
adssx
#386
Do you have a link to the source?
A quick search on Google Scholar points to… zero evidence in favor of PF4 for PD. Do you have some sources?
1 Like
Satchel
#387
You have to read a couple of papers to put it together, -which I will list. In a nutshell Klotho via PF4 abrogates alpha-synuclein affects on cognition to 70% of normal. - but does not help motor difficulties (which they have established treatments for). I have to search again but i had found a clinical trial a few months ago- I will hunt it down as well. Basically why bother with Klotho when an established treatment is already approved now-all be it for a different disease process.
https://www.nature.com/articles/s43587-023-00468-0
Quote from Dena Dubal talk with Peter Attia -“PF4 to mice as a shot in the belly (just like they had given klotho), and found it enhanced cognition in young mice, and in an aging mouse it reversed cognitive defects ‒ it was totally remarkable”
1 Like
Satchel
#388
The top link doesn’t seem to connect so here is the title
Peripheral Elevation of a Klotho Fragment Enhances Brain Function and Resilience in Young, Aging, and α-Synuclein Transgenic Mice
adssx
#389
That’s the first paper in mice?) We don’t even know if alpha α-synuclein causes PD, same issue with beta amyloid in AD…)
What does this mean? There’s no disease-modifying treatment for motor symptoms in PD. There’s only levodopa that reduces the tremors but after 5 years it gives you dyskinesia that is even worse.
It does seem aberrant splicing is part of PD.
I started playing around with coversations with 01 about splicing, cancer, PD etc.
I do this in the dark because it enables my superchiasmatic nucleus to think it is the end of the day (which it is) whilst still processing information.
What I really need to do is to put together something from o1 where I have checked the references and tracked things down to avoid hallucinations that looks at aging, cancer, diabetes, PD and aberrant splicing.
What seems to be the case is that acetyl-CoA shortage causes both splicing changes and otherwise long genes not to be transcribed.
This is quite rational, but appears to drive the phenotypes of aging which includes PD. It would be good to do some biohacking trials with younger PD sufferers to see if this* can fix it.
*this being increasing cytosolic acetyl-CoA through a range of biohacking techniques.
1 Like
adssx
#391
And klotho and/or PF4 would fix that @John_Hemming? (I don’t know enough about klotho…)