Just as with rapamycin, this treatment seems to provide longevity benefits without changing epigenetic age measures:
Aging-related decreases in cardiac and skeletal muscle function are strongly associated with various comorbidities. Elamipretide (ELAM), a novel mitochondria-targeted peptide, has demonstrated broad therapeutic efficacy in ameliorating disease conditions associated with mitochondrial dysfunction across both clinical and pre-clinical models. Herein, we investigated the impact of 8-week ELAM treatment on pre- and post-measures of C57BL/6J mice frailty, skeletal muscle, and cardiac muscle function, coupled with post-treatment assessments of biological age and affected molecular pathways. We found that health status, as measured by frailty index, cardiac strain, diastolic function, and skeletal muscle force, is significantly diminished with age, with skeletal muscle force changing in a sex-dependent manner. Conversely, ELAM mitigated frailty accumulation and was able to partially reverse these declines, as evidenced by treatment-induced increases in cardiac strain and muscle fatigue resistance. Despite these improvements, we did not detect statistically significant changes in gene expression or DNA methylation profiles indicative of molecular reorganization or reduced biological age in most ELAM-treated groups. However, pathway analyses revealed that ELAM treatment showed pro-longevity shifts in gene expression, such as upregulation of genes involved in fatty acid metabolism, mitochondrial translation, and oxidative phosphorylation, and downregulation of inflammation. Together, these results indicate that ELAM treatment is effective at mitigating signs of sarcopenia and cardiac dysfunction in an aging mouse model, but that these functional improvements occur independently of detectable changes in epigenetic and transcriptomic age. Thus, some age-related changes in function may be uncoupled from changes in molecular biological age.
Open access paper:
https://onlinelibrary.wiley.com/doi/10.1111/acel.70026
Relacionado:
Elamipretide (also known as SS-31, MTP-131 and Bendavia)[1][2] is a small mitochondrially-targeted tetrapeptide (D-Arg-dimethylTyr-Lys-Phe-NH2) that appears to reduce the production of toxic reactive oxygen species and stabilize cardiolipin.[3]
Stealth Peptides, a privately held company, was founded in 2006 to developintellectual property licensed from several universities including elamipretide; it subsequently changed its name to Stealth BioTherapeutics.[4][5]
As of November 2017 Stealth had obtained an orphan designation in the US for use in mitochondrial myopathy and had started a Phase III trial in that indication.[2] As of January 2020, trial expectations were not met.[6]
https://www.sciencedirect.com/topics/pharmacology-toxicology-and-pharmaceutical-science/elamipretide
3 Likes
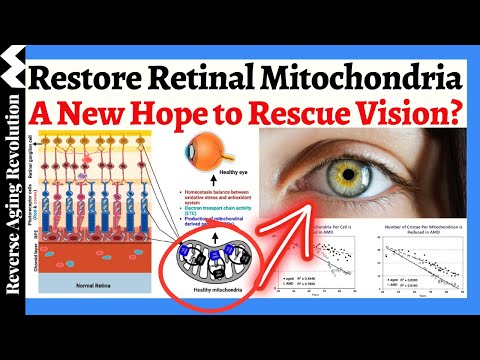
Restoring Retinal Mitochondria - A Revolutionary Approach To Improve Your VISION
Is a revolutionary peptide therapy targeting mitochondria the breakthrough we’ve been waiting for? In this video, Ms. Reenie McCarthey, CEO of Stealth Biotherapeutics delve into the exciting advancements from a Phase 3 clinical trial exploring the potential of this novel approach to vision restoration in Dry Age-Related Macular Degeneration (AMD)
We’ll break down:
• The critical role of mitochondria in retinal health: Discover how mitochondrial dysfunction contributes to Dry AMD and why targeting them is crucial.
• The science behind the peptide therapy: Learn how this innovative treatment works by interacting with cardiolipin to restore mitochondrial structure and function.
• Key findings from the Phase 2 trial: We’ll discuss the encouraging results, including improvements in low-light visual acuity and the protective effect on photoreceptors.
• Updates on the upcoming Phase 3 trials: Get the latest information on the global trials, including enrollment timelines and what to expect.
• The potential for vision improvement: We’ll explore how this therapy aims to not just slow progression but potentially improve vision for Dry AMD patients.
• Why this approach is different: We will cover how targeting the mitochondria is getting ahead of the disease progression, instead of treating the end result.
1 Like
Wow, 40mg daily via subcutaneous injection. That is a very high dose compared to what is available on the “gray market” (known as peptide SS-31), which is typically 30mg vials that sell for $300 (give or take) for a kit of 10 vials (which comes along with caveats of purity, potency, possible contaminants, etc). So about $1,200 per month.
Perhaps much lower doses started much earlier might work as an AMD preventative vs a higher dose being required for treatment of advanced disease?
Mitochondria-Targeting Abasic Site-Reactive Probe (mTAP) Enables the Manipulation of Mitochondrial DNA Levels
https://onlinelibrary.wiley.com/doi/10.1002/anie.202502470
Summary about how this works:
Chemical Shield Stops DNA Damage from Triggering Disease–’A Paradigm Shift’
In lab tests as well as studies using living cells, the probe significantly reduced mtDNA loss after lab-induced damage mimicking exposure to toxic chemicals such as nitrosamines, which are common environmental pollutants found in processed foods, water, and cigarette smoke.
2 Likes
I’ve contacted Stealth Bio to inquire about this phase of the trial since I have dry AMD. Not sure if the phase was discontinued, but no reply from them or 2-3 of the retina clinics I’ve contacted. One clinic claimed they withdrew.
1 Like